Cryotherapy for Prostate Cancer
Cryotherapy (or cryosurgery / cryoablation) is a minimally-invasive effective treatment for prostate cancer with fewer side-effects than surgery or radiotherapy.
What is focal therapy?
Focal Therapy involves treating 1-2 areas of cancer in the prostate so that less tissue is damaged. This means the cancer is treated but without causing collateral tissue damage to nerves, muscles, bladder and back passage. Recovery is quicker than radical surgery or radical radiotherapy as well. Focal therapy can be carried out using different energies which ablate (or destroy) tissue precisely. One of these is cryotherapy.

What is Cryotherapy?
Cryotherapy for prostate cancer is a minimally invasive treatment that can be used to ablate or destroy areas of tissue within the prostate gland. It can also be used to treat other cancers in other places in in the body. Cryotherapy uses extremely low temperatures, as low as -40 to -100 degrees centigrade, that free areas of the prostate gland. Only the cells within ablative freezing temperatures are destroyed whilst tissue elsewhere in the prostate or surrounding the gland are not affected.
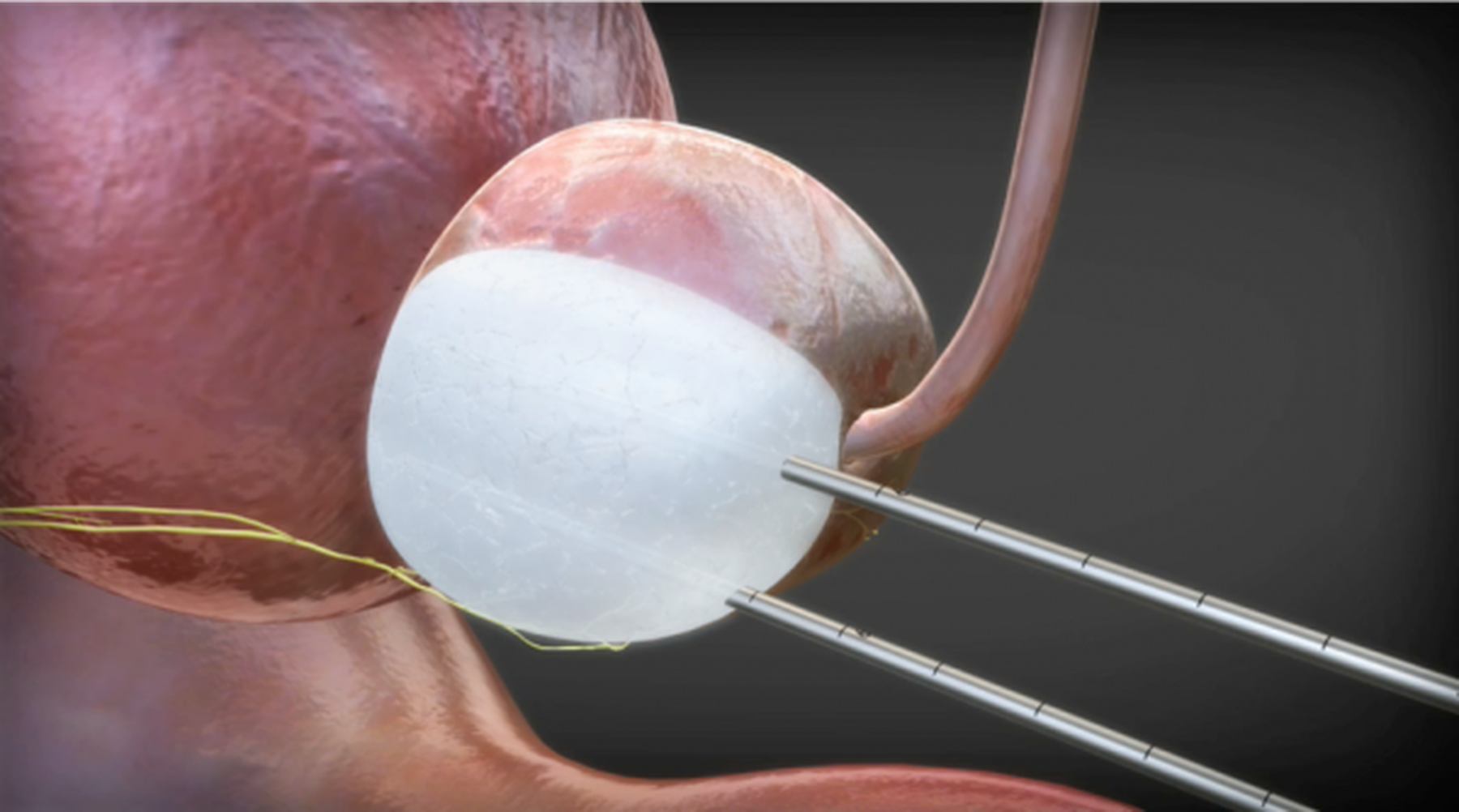
With cryotherapy for prostate cancer, it can be used to target individual areas of cancer (with a margin of normal tissue) with multiple needles placed in and around the cancer area(s) so that a region of the prostate can be treated. Cryotherapy for the treatment of prostate cancer can be used to treat the entire prostate or it can be used to treat areas of prostate cancer, and usually no more than two areas. Whole-gland cryotherapy is less commonly used nowadays but in some circumstances can still be used such as in patients after radiotherapy who have a recurrence or in trials such as the IP2-ATLANTA study that Professor Ahmed is currently recruiting patients with metastatic prostate cancer to.
Focal cryotherapy which is the use of cryotherapy to treat individual areas of prostate cancer within the gland is now much more commonly used. The concept of focal cryotherapy for prostate cancer is similar to breast lumpectomy. It carries fewer side effects compared to radical whole gland treatments like radiotherapy or surgical removal of the prostate (prostatectomy) and recovery back to normal activity is also sooner.
Is cryotherapy used instead of surgery (prostatectomy) or radiotherapy for the treatment of prostate cancer?
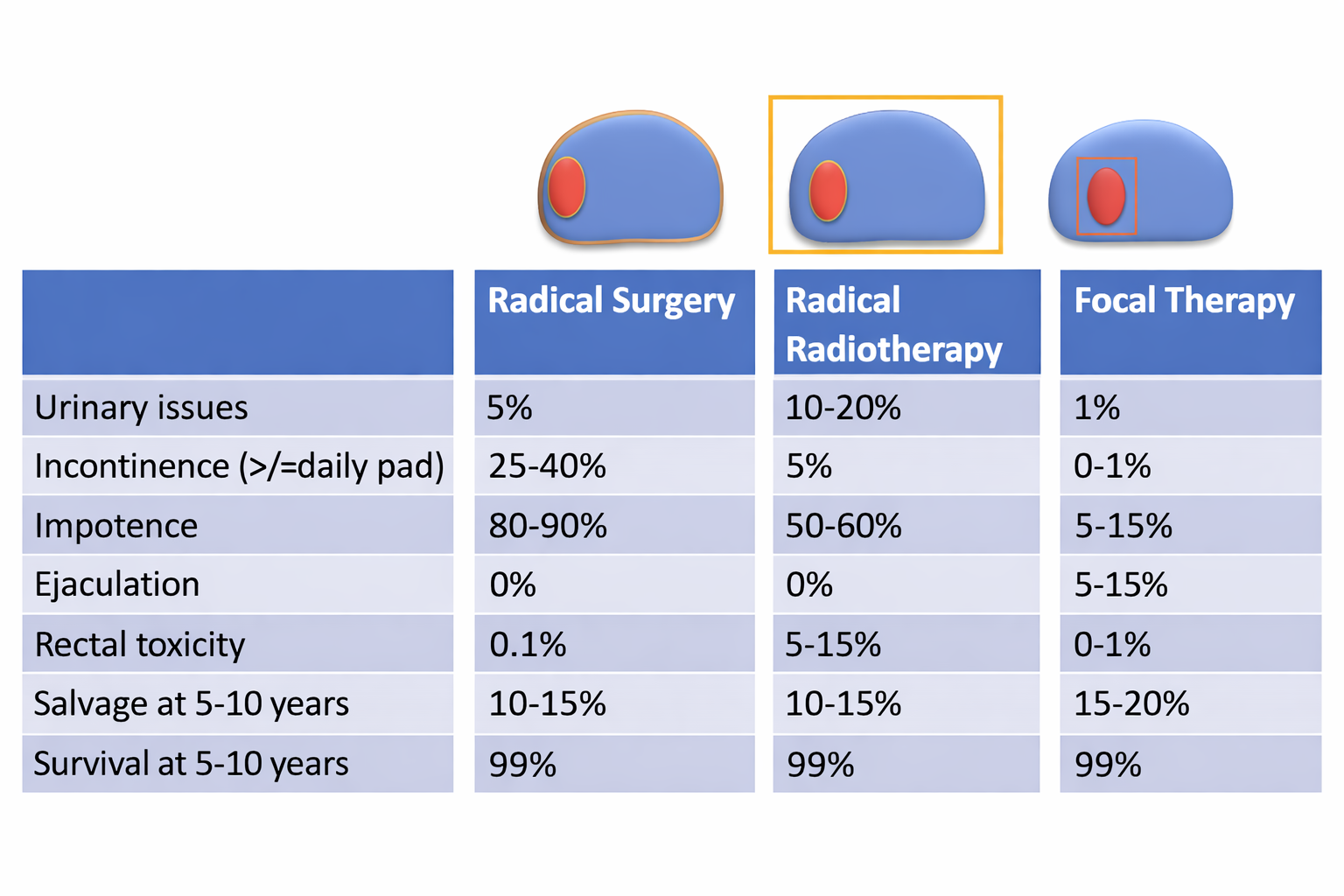
In patients who are advised to have an active form of treatment because the cancer would otherwise progress or grow if they didn’t have treatment can have cryotherapy provided the cancer is limited to one or two areas within the prostate. It is also important that the prostate cancer has not shown any signs of distant spread or what is called metastases. The vast majority of patients who were diagnosed with prostate cancer that has shown no sign of spread are offered whole-gland radical radiotherapy or prostatectomy surgery which involves removing the entire prostate. These treatments are very effective and are known to improve cancer control and survival in the long term but that cancer control and survival in the long term in the majority of cases is a small benefit over monitoring the cancer.
On the flip side, when you treat the whole prostate with radiotherapy or surgery one can cause damage to collateral tissue which surrounds the prostate such as neurovascular bundles which supply the penis for erections and nerves and muscles which control urine flow and stop leakage. The water passage, which we call the urethra, runs through the prostate and this can get damaged and cause urinary symptoms such as frequency, urgency and discomfort; the bladder is also very close to the prostate and this can get damaged and cause again lead to frequency, urgency and discomfort when passing urine. Finally, the back passage is literally millimetres away from the prostate and with radiation especially some of the radiation can affect the back passage or the rectum and this can lead to back passage symptoms such as discomfort, bleeding and loose stools.
In summary, following radical prostatectomy (surgical removal of prostate), the long-term risk of urine incontinence requiring at least one pad a day is between 15% and 25% with about 5% to 10% of patients requiring more than one pad a day. These percentages are higher in the first 6 to 12 months after recovery. The long-term risk of erectile dysfunction depends on the degree of nerve preservation that is performed but on average the range of erectile dysfunction varies between 30% and 60% of patients. All patients will lose ejaculation and in those patients that get erections back many will need to use medication or injection or vacuum treatment. About 5% of patients will get a narrowing in the water passage where the bladder and the urethra has been joined at what we call the anastomosis site.
In summary, following radical radiotherapy, the long-term risk of urine incontinence in which leakage occurs when you cough or sneeze or lift something is much lower at less than 5%. However, other types of urinary problem can occur such as urgency, frequency and getting up at night as well as discomfort when passing urine due to the radiotherapy causing an irritation of the bladder and the urethra. Some patients will need to use pads on a daily basis in order to stop the leakage affecting them when they get strong urge to pass urine. Erectile dysfunction after radiotherapy in the long-term is affected in about 50% of patients with most not able to have ejaculation, and again depends on the function before treatment. Radiotherapy in addition can cause about 5% of patients to have severe bowel symptoms such as bleeding, discomfort and loose stools with another 5% to 10% suffering mild-to-moderate bowel symptoms. Brachytherapy, which is a form of radiotherapy involving inserting radioactive pellets into the whole prostate causes very types and levels of risk to urinary, sexual and bowel function but is carried out differently to external beam radiotherapy. Evidence so far has not supported the rationale that stereotactic radiotherapy or proton radiotherapy leads to fewer side-effects or is more effective, although fewer doses and sessions of radiotherapy are required.
Cryotherapy for prostate cancer treatment is therefore an alternative in those patients that are eligible, instead of whole-gland radical surgery or radiotherapy. It is not for every single patient who is diagnosed with prostate cancer and the detailed eligibility criteria are discussed below although a detailed discussion of your circumstances is very important with an expert in focal therapy. Doctors who do not carry out focal therapy can sometimes mistakenly state patients under their care are no eligible when they so always ask to speak to or see a focal cryotherapy specialist.
Is cryotherapy used instead of active surveillance for prostate cancer?
Patients who have prostate cancer which has not spread and is low risk or a small amount of medium risk cancer are suitable for active surveillance. This involves monitoring the prostate cancer with regular PSA blood tests, clinical exam, MRI scans and sometimes biopsy. The strategy of active surveillance is very safe for these patients. This is because low-risk prostate cancer in the majority doesn’t grow or spread and medium risk prostate cancer grows very slowly when it is a small amount. So, generally, we would not advise any kind of active treatment as an alternative to active surveillance for patients who have proven low-risk prostate cancer on a high quality multiparametric MRI and image fusion targeted and systematic prostate biopsy. Patients who have medium risk prostate cancer have the option of active surveillance or focal cryotherapy or focal HIFU, or if they wish, surgery or radiotherapy to treat the entire prostate. The choice is up to the individual patient in discussion with his doctors.
There are some patients who have low risk prostate cancer who psychologically would not be able to cope with monitoring prostate cancer even after being reassured that it is indolent and carries very little chance of causing an impact on their life expectancy or quality of life. There are also some patients who on active surveillance do not show any signs of progression of the cancer but find the burden of active surveillance with repeated tests and MRI and potentially biopsies too much. At some point they decide to have active treatment. Last, a small number of patients will show progression of the cancer whilst they are on active surveillance and need to consider active treatment. The majority of all of these patients are eligible to choose from all of the active treatment options including focal therapy using cryotherapy or HIFU.
What are the side effects from cryotherapy for prostate cancer treatment?
As with any treatment there are always some early side effects and some late side effects. Professor Ahmed will discuss these with you and will also provide an information sheet that will detail these.
Immediately after the cryotherapy treatment for your prostate cancer most patients will feel tired and lethargic from the anaesthetic and then up to 6-8 weeks will feel ongoing tiredness as a result of the body’s reaction and healing process to the area that has been damaged by the cryotherapy treatment. This is natural and what we would expect when the body heals itself. For the first few weeks after cryotherapy you will get symptoms of urgency, getting up at night, frequency of urination as well as some discomfort when you pass urine. Because the prostate is swollen and inflamed the flow of urine may also be slower. On average, symptoms take about 6 to 8 weeks to settle down but in some patients can settle down quicker, and in some patients can take up to 3 months to fully settle. Most patients are able to get back to a desk job after two or three weeks of resting at home and during this period of time there is no need to be bed-bound or housebound and you should try and get back to normal non-strenuous activity as soon as possible. We advise avoiding strenuous activity or anything that puts pressure on the abdomen for the first 4 weeks and then returning back to those kinds of activities slowly over the following 2 or 3 weeks. Some patients will pass blood occasionally and also occasional blood clots or some tissue or debris within the urine. This is also expected because the body is expelling the dead tissue similar to how a scab forms on a cut on the skin; except that this scab is within the water passage where the prostate resides. Some patients will develop a urine infection and require antibiotics and a small number of patients develop an infection within the testicle called epididymo-orchitis. Very rarely will an infection lead to admission to hospital to have antibiotics through the vein for a few days.
There are other complications which can occur from any kind of operation within the pelvis such as chest infections or blood clots within the leg which can very rarely occur and then cause blood clots in the lung called pulmonary embolus. For that reason, you will be given compression stockings to wear as well as have active compression during the operation and at the end of the operation, provided there are no contraindications, you will also be given a blood thinning injection whilst under anaesthetic.
In the long-term, the risk of incontinence of urine leading to at least wearing one pad a day is about 1 in 100. In some patients, where the tumour is closer to the sphincter muscle that stops leakage of urine, the risk can go a little higher and Professor Ahmed will specifically advise you about that. The risk of erectile dysfunction after focal cryotherapy for prostate cancer varies between 5% and 20% depending on the amount of tissue that is treated and depending on the function before the cryotherapy. So, in patients with good baseline function the risk is about 5% to 10% and patients with not so good baseline function the risk is closer to 15% to 20%. About quarter of patients will need to use medication to help with erectile function and many of these patients continue on medication long term. If two areas of the prostate on both sides are treated the risk of erectile dysfunction can double. The risk of dry orgasm or not having any fluid on ejaculating is on average 50%. In some patients who have treatment to both sides of the prostate or where the focal treatment is close to the middle of the prostate where the two tubes that deliver the fluid join together will have close to 100% risk of dry orgasm. Even in those patients who have some fluid the volume will be lower because some of the tissue and one tube inside the prostate will have been damaged. If a patient does get ejaculation fluid, then there’s a good likelihood that there will be viable sperm within the fluid and he and his partner should regard themselves as being able to conceive naturally and should take alternative contraception precautions if necessary. If dry orgasm does occur there is very little that can be done to reverse this at the moment.
The risk of rectal or back passage or bowel problems in the long term is rare. Some patients will get some bowel upset such as constipation in the first few weeks after the cryotherapy though this tends to settle down. Rarely, in about 1 in 1000 to 1 in 700 patients back passage damage can occur in the form of a recto-urethral fistula. This means a hole has been created in the back passage which connects with the water passage. This risk is the same as the risk of surgery causing a tear in the back passage during the operation or radiotherapy causing a fistulous tract. If a fistula does occur, then a long-term catheter will need to be placed to divert the urine and allow the body to heal the tract up on its own over 3 to 6 months. If the tract does not heal then an operation will be needed.
Finally, narrowing in the water passage can sometimes occur either due to retained necrotic or dead tissue which then needs to be removed using a cystoscopic operation under general anaesthetic or the healing process leads to a scarring and a stricture (narrowing) in about 1-2%.
How effective is cryotherapy in treating prostate cancer?
About 20% to 30% of patients over a 6 to 7-year period will require a second cryotherapy session to be applied to the same area that was treated at the beginning. The exact risk depends on the risk of the cancer at the time of the first treatment. We regard the second session in these patients as part of the focal therapy strategy. Most patients regard this as an advantage, and we ask patients who choose focal therapy using cryotherapy to consider that there is a chance that the cryotherapy treatment may need to be repeated. So far about 3% to 5% of patients over the same follow-up period on average develop new tumours in the untreated tissue which can either be treated with further cryotherapy or other forms of focal therapy or at that point these patients can decide to have surgery or radiotherapy. About 5% to 10% of patients overall despite two treatments of cryotherapy will require surgery or radiotherapy due to a recurrence. The chance of cancer spreading is rare and so far, the chance of cancer-related death is also very rare and similar to following surgery and radiotherapy.
In the UK, almost 1000 patients have been treated with focal cryotherapy (up to 2020) since 2007. The majority (80% to 90%) have medium-risk prostate cancer and about 10% to 20% have high-risk cancer. When we have compared our outcomes to surgery and radiotherapy, we found that focal therapy (using HIFU or cryotherapy) has very similar cancer control over a 6 to 7-year period (see here). However, we do not have long term data of average 10 to 15-year follow-up and do not have clinical trials called randomised control trials in which the patients are allocated by a computer program randomly to either have focal therapy or radical treatment.
Is cryotherapy treatment for prostate cancer approved by NICE in the UK?
Focal cryotherapy is currently approved by the National Institute for Health and Care Excellence (NICE) in the UK, through the interventional procedures guidance route and NICE IPG423 which can be found at the following NICE Cryotherapy guidance. In this guidance NICE has asked urologists who carry out focal cryotherapy to make sure they are fully trained and approved to carry this out, that they inform the hospital they are doing it, that all cases are discussed at a cancer multidisciplinary team meeting for suitability, that patients are fully informed in writing as well as verbally about the pros and cons of the treatment, and finally that they submit their data to a national registry which will collect outcomes of the treatment in the long term. Professor Ahmed is the Chair of Focal Therapy UK which is the user group of focal therapy urologists which oversee the national registries for focal cryotherapy and focal HIFU called ICE and HEAT. These registries have reported their data periodically in peer-reviewed high-impact journals. Focal Therapy UK can be found at this Focal Therapy UK with a list of approved urologists who carry out focal HIFU and focal cryotherapy.
What happens on the day of cryotherapy for prostate cancer treatment?
On the day of treatment, you will be met by the administrative and nursing team who will carry out an update of your medical file to make sure that there are no other issues that the anaesthetist or the surgeon need to consider. You will then have an opportunity to meet with the surgical team who will go through formal written consent and answer any further questions that you have. The process of consent is very much one in which the it starts on the first consultation and is reinforced on subsequent consultations and in written format with patient information sheets and information such as Professor Ahmed’s website, as well as the final written consent form which should carry a summary of the treatment and it’s side effects and complications.
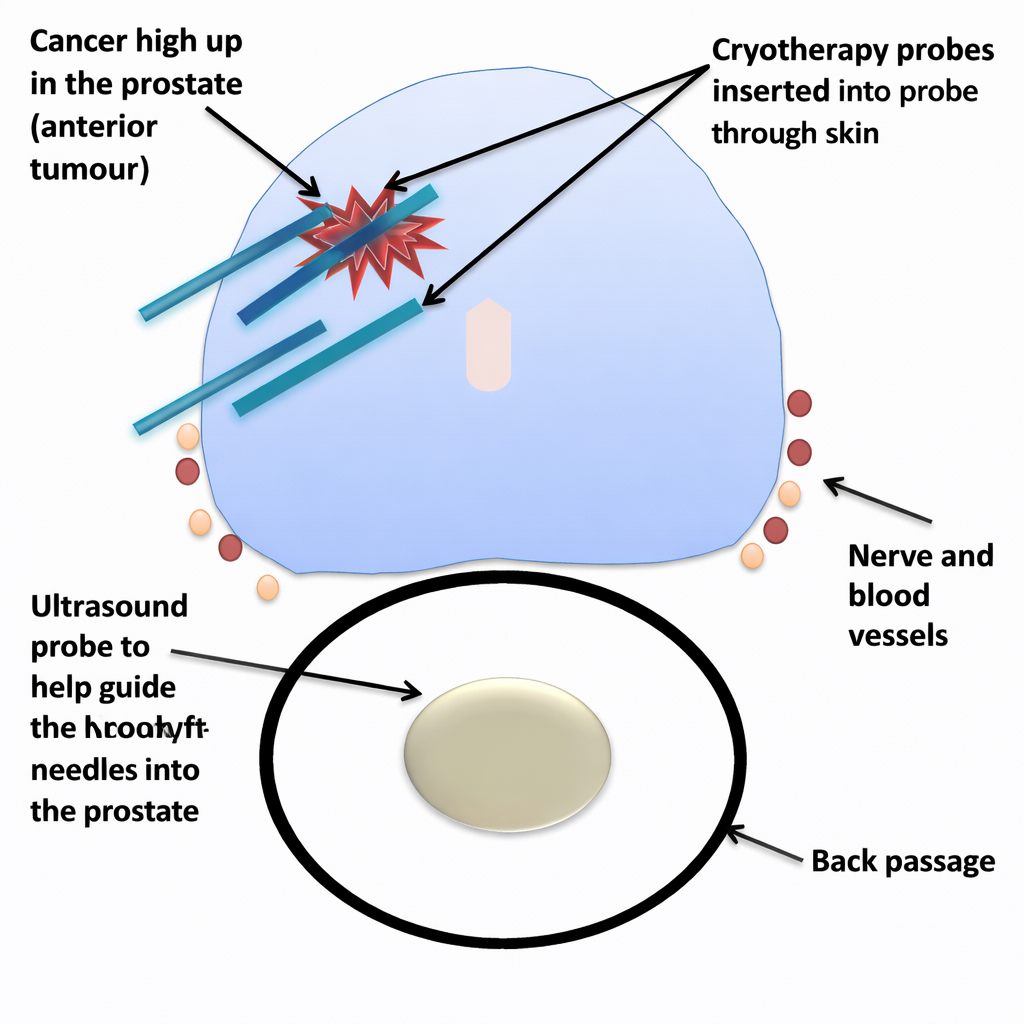
Cryotherapy for prostate cancer is normally carried out under a general anaesthetic although occasionally a spinal with deep sedation can also be carried out. The critical aspect is that the patient must be entirely still because this is precision surgery and any move of the patient can make the cryotherapy damage other parts such as the rectum or the sphincter muscle or miss areas of cancer inadvertently. The treatment involves an ultrasound imaging probe that is inserted through the back passage which then scans the prostate using ultrasound waves; this is similar in size to what was used during your prostate biopsy. The surgeon will then plan the treatment and insert the cryotherapy probes through the skin behind the scrotum and in front of the back passage – what is commonly referred to as the transperineal route.
The needles that are inserted into the prostate are similar in diameter and length to the biopsy needles. The cryotherapy machine allows tweaking and refinements of the treatment constantly throughout the delivery of treatment and allows the surgeon to raise or lower the temperature depending on what changes they are seeing on the ultrasound images and also by monitoring the temperature using thermocouples inserted near the tumour and at other points within or outside the prostate. This refinement has been shown in studies to offer a significant advantage in effectiveness and safety of the treatment.
As a result, expertise and doing lots of cases is critical to doing cryotherapy. Professor Ahmed is one of the most experienced cryotherapy urologists globally having performed hundreds of cases over the last decade, and has taught many cryotherapy surgeons the technique around the world. His cryotherapy practice is one of the largest globally.
The whole procedure can take between 1.5 and 2.5 hours depending on how large the prostate is, and how large the tumour and the overall area that is being treated. Once you are awake and recovering from the anaesthetic you will feel quite groggy and drowsy and you may not remember much about what the surgeon has told you about the operation. Often a member of the surgical team will speak to you the following day. If you leave a next-of-kin number, we can call them to inform them how the operation went on the same day so that they’re not worrying.
A catheter will be inserted through the urethra which is inside the penis at the end of the operation or at the beginning. Catheters are soft plastic tube that have a balloon at the end of it in order to stop it from falling out. Sometimes the catheter is inserted through the skin below the belly button (suprapubic catheter) if the surgeon believes that this would be a better form of inserting a catheter. This will be discussed by the surgical team.
The catheter can make you feel like you want to pass urine because of the balloon at tip of the catheter stimulating the pressure sensors within the bladder. The bladder tends to settle with time, but this sensation may not fully go away until the catheter is removed 1 to 2 weeks later. Most patients will have a catheter removed after 7 to 9 days, but if you have had cryotherapy for cancer after radiotherapy or if you have had a large amount of tissue treated or if there has been a lot of swelling of a large prostate than the catheter may need to stay in for 10 to 14 days because removing it earlier would risk you not being able to pass urine on the day the catheter is removed. When the catheter is removed there is about a 1 in 20 chance that you may not be able to pass urine. In this instance, we prefer that you carry out clean intermittent self-catheterisation (CISC), so that you can try and pass urine normally and then stop with the self-catheterisation once you are passing urine well. This avoids you needing to have a catheter in place all the time which can be uncomfortable and increase the risk of infection.

What is the follow-up after cryotherapy treatment for prostate cancer?
At 1 to 2 weeks you will have the catheter removed by one of the nursing team. You will have a consultation booked either in person or remote telephone consultation at the 3-month stage although Professor Ahmed will be able to speak to you before this if there are any issues or concerns greatly affecting you. At the 3-months stage we ask that you have a PSA either at the Cromwell Hospital or with your GP and if you have it with your GP to send the result in before the consultation.
Most patients will see a drop-in their PSA of anywhere between 20% and 90% from the baseline value. The amount the PSA drops depends on the amount of inflammation still present in the prostate because inflammation of the untreated tissue can make the PSA go up and also the amount of tissue that was a ablated and the amount that the specific cancer in your case was contributing to your baseline PSA value. A small minority of patients will see no drop-in the PSA or even an increase in the PSA at the 3-month stage and in the vast majority of these cases this is nothing to worry about and is due to residual ongoing healing of the prostate which causes inflammation and the untreated prostate tissue to secrete much more PSA than it normally does. In this instance, Professor Ahmed will advise you to take another PSA 2 to 3 months later and at that point take some anti-inflammatories beforehand for about a week in order to calm down any residual information that might still be present before having the PSA blood test.
If the PSA has shown a good response, then we normally wait until the 12-months stage to do another PSA and an MRI. It can take 6-months and in some patients 9 months for the healing process to fully settle down and doing an MRI any earlier means that the inflammation or necrotic tissue still present can lead to an inaccurate MRI test. A small minority of patients will have signs of residual cancer at the 12-month stage and at that point the vast majority will have a further cryotherapy. If there is no sign of residual disease and no new cancer lesions occurring at the 1-year stage, then you will continue on 6 monthly PSA blood test follow-up with yearly reviews by Professor Ahmed. Only if the PSA rises consistently will another MRI scan be needed. Often the PSA will fluctuate from one test to another and provided that there is no overall trend upwards this is normal. If there is a rise in the PSA from one test to another it is worth repeating it two or three months later with a course of anti-inflammatories like ibuprofen or naproxen (provided it is not contraindicated in your case) or antibiotics in order to settle down any inflammation that might be leading to the fluctuations. If at any point, there is sign of recurrent cancer in the treated area then the majority of patients will have further cryotherapy which again will treat just the area of cancer not the whole prostate.
Can prostate cancer return after Cryotherapy?
Yes, prostate cancer can return after cryotherapy in a small proportion of patients. No cancer treatment, including cryotherapy, offers a 100% guarantee of permanent remission. Patients should continue regular monitoring through blood tests, imaging scans, and clinical evaluations to check for signs of cancer recurrence. For every 100 patients who have focal cryotherapy, about 20 patients need to have the treatment repeated within 5-10 years. Of these 20 patients, about 5-7 then need to have another form of treatment. These rates of treatment are not too dissimilar to radical prostatectomy or radical radiotherapy.
If I have cryotherapy can I have cryotherapy again?
If there is residual cancer or a recurrent cancer on follow-up the majority of patients can have cryotherapy again to that area. Some patients have new areas of cancer in untreated tissue and in these instances again many choose to have focal cryotherapy to those individual new areas. At any point a patient may choose any form of surgery or radiotherapy.
If cryotherapy doesn’t work can surgery or radiotherapy still be done?
If cryotherapy for prostate cancer doesn’t work, then surgery and radiotherapy can still be carried out. Surgery is a little bit more difficult because there is going to be scar tissue around the area where the cryotherapy was carried out, but the majority of the prostate will not be affected. The majority of expert surgeons who have done this type of surgery have said it is not as difficult as surgery carried out for patients who have previously had radiotherapy to the entire prostate and have had a recurrence. The risk of erectile dysfunction is higher with salvage surgery but the risk of incontinence of urine does not seem to be higher compared to patients who have had surgery right at the outset.
Radiotherapy can be done as well after cryotherapy for prostate cancer and again in expert hands is somewhat straightforward and doesn’t seem to add additional urine incontinence or erectile dysfunction risk compared to patients who have radiotherapy upfront.
It is important to remember that about 10% to 15% of patients who have surgery will go on to have a recurrence requiring radiotherapy or hormones or both. About 10 to 15% of patients who have radiotherapy will go onto have hormones or surgery or focal treatment (cryotherapy or HIFU) due to recurrence. Unfortunately, the nature of cancer is that no treatment is 100% effective and some patients will unfortunately have a risk of failure or recurrence. The key thing to note in these areas of discussion is that survival from a cancer-specific perspective is the same between all of these treatments.
Who is suitable for cryotherapy for prostate cancer?
The vast majority of patients that are treated with cryotherapy for prostate cancer have medium (or intermediate) risk prostate cancer. This means that the cancer is contained within the prostate and has a Gleason score of 3+4=7 or 4+3=7. This is commonly now referred to as ISUP Grade Group 2 or 3. Provided that one or two areas can be treated with cryotherapy and there are no other contra-indications, this is the optimal group of patients. High risk disease which is small or just touches the capsule (edge) of the prostate can also be treated with cryotherapy; in the UK about 10% of cryotherapy cases are high risk.
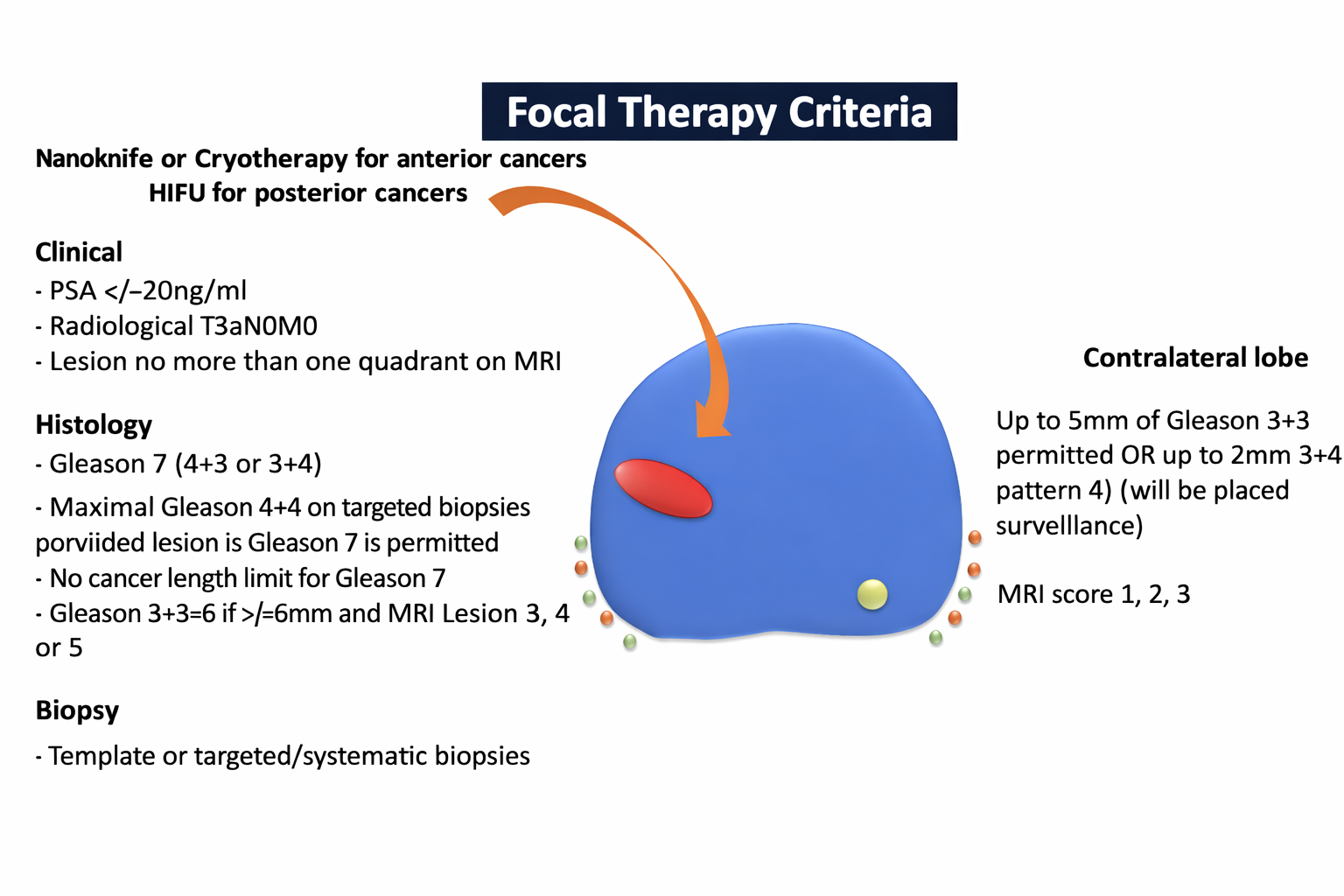
In a lot of patients, there is one main tumour which is called index tumour and we know from a lot of biological studies that this index tumour is what drives the natural history and progression of the cancer. Many patients will have tiny little bits of low-risk disease which is not visible on MRI which has a Gleason score of 3+3=6 and is commonly regarded as indolent disease which often occurs in about one in three of the general male population above the age of 50. Cryotherapy commonly involves treating the index lesion and monitoring the areas of secondary indolent low-risk disease because these areas on the whole do not progress and would be picked up on monitoring if they did. The reason not to treat every area of disease that is found regardless of risk is that the more tissue treated the closer the treatment comes to whole gland surgery and radiotherapy and therefore the advantages of lower side effects and lower complications start to disappear.
In some patients who have disease suitable for cryotherapy, but it is in a place or in a gland that is difficult for cryotherapy to be technically delivered, Professor Ahmed might advise focal HIFU or Nanoknife as a second or first therapy. This is because the tumour is lower down in the prostate in the peripheral zone or close to the sphincter muscle or the urethra.
Finally, patients who have low risk disease who do not want to or cannot tolerate active surveillance or progress on active surveillance. It is important however, to strongly consider that in most instances, we prefer not to treat even with focal cryotherapy just because of heightened anxiety and would be disappointed if surgery or radiotherapy was being used in this way as there is likely to be little cancer benefit.
Patients who have high volume, low risk prostate cancer on biopsy where the tumour is visible on the MRI will have a higher risk of progression on active surveillance and often in these patients, active treatment is offered. Focal therapy is an option in these cases.
To hear Professor Ahmed talk about the prostate cancer pathway and focal therapy for prostate cancer, see here.
Further information
NHS referrals
Professor Ahmed works at Imperial College Healthcare NHS Trust. You are entitled to ask for a referral for an opinion about whether you might be suitable on the NHS.
Your GP can make the referral through the choose and book electronic referral system and choose the 2ww-Prostate folder. Your consultant team can also make the referral directly to Imperial using the email address:
imperial.prostate@nhs.net.
Alternatively a letter can be sent to:
Professor AhmedCharing Cross Hospital
Fulham Palace Road
London, W6 8RF
Private referrals
For private referrals, Professor Ahmed works at Cromwell Hospital and Imperial Private Healthcare. Self-pay patients do not need a referral to consult Professor Ahmed and there are competitive self-pay packages offered. Insured patients should check with their insurer. Please use the online booking portal or email the team on london.prostate@gmail.com.
Related Treatments
-
HIFU for prostate cancer treatment
A minimally invasive treatment that can be used to ablate or destroy areas of tissue within the prostate gland with a low risk of side-effects.
-
Targeted transperineal prostate biopsy
This procedure involves fusing ultrasound probe images of the prostate with the multiparametric MRI of the prostate taken prior.
